|
Sharing of one valence electron pair results in a single bond between two constituent atoms inside a molecule, sharing of two pairs ends up in a double bond and that of three pairs forms a triple bond. Note: Usually the elements obtain their octet configuration due to the chemical bond formation.Ĭovalent bonds are formed via the electron sharing process where atoms of the same or different elements come together to share electron pairs among themselves. We usually witness this sort of bond formation in a molecule formed by a metal and a non-metallic element. There is the formation of a positively charged cation and a negatively charged anion which come together due to electrostatic attractive forces. Thus, while one atom loses an electron(s), the other atom gains an electron(s). Ionic bonds are formed via an electron exchange process where an atom of a comparatively electropositive element transfers electron(s) to the comparatively electronegative one. 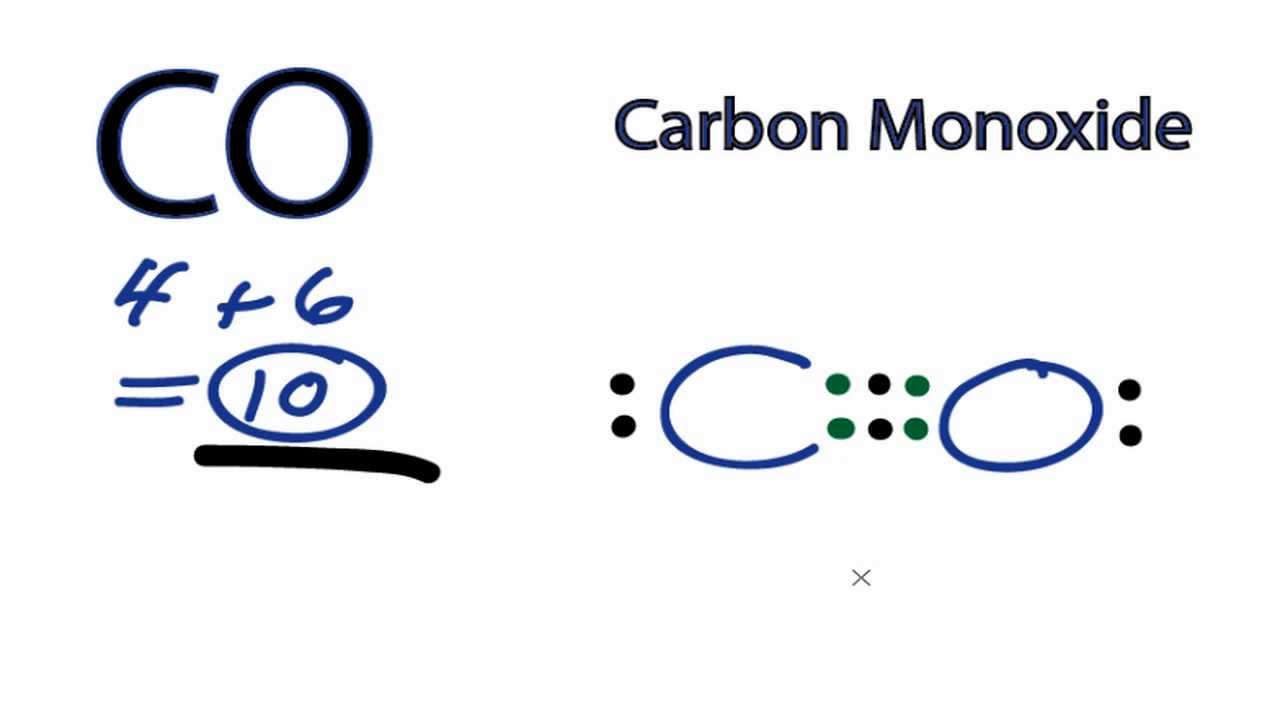
However, in this article, we will restrict our discussion to two major types: Ionic and Covalent. This chapter deals with various types of chemical bonds. The electrons present in the outermost shell, also known as valence electrons, have a tendency to take part in the bond formation as a result of which we get several products. It deals with the attractive nature of atoms to combine with one another and form different types of molecular compositions.Ītoms have negatively charged subatomic particles called electrons around the nucleus. 
To do so, we have to study chemical bonds and their types and how they are formed in order to have a crystal clear idea.Ĭhemical bonding is one of the most significant aspects of chemistry. Here, we can see a number of chemical reactions that lead to the formation of this product:īefore we can discuss whether CO2 is ionic or covalent, we have to first understand the nature of chemical bonding inside the molecule. We can produce and manufacture this compound via several means. It bears a colorless and odorless gaseous appearance which is capable of dissolving in water to form carbonic acid H2CO3. Let us now understand it in detail:īefore that, let us look at some of the tidbits about the famous molecular composition.ĬO2 has a molecular weight of about 44.009 g/mol. The above explanation is just a gist of the topic we have dealt with. 
Oxygen and Carbon both are present in the second period and therefore achieve the outermost shell configuration of the noble gas Neon. Doing so, both C and O attain octet fulfillment. Other than this, here in each bond of C and O, the atom of carbon shares four electron pairs with oxygen forming a double bond. In a CO2 molecule, between C and O, the difference is around 0.89, so we can call this a covalently bonded molecule. So, today, in this article, we will discuss Carbon Dioxide – whether it is ionic or covalent in nature.Ī compound is said to be covalent when it has an electronegativity difference between its bonded atoms to be less than 1.8-2. 
Now, you might be wondering about the nature of bonding in CO2 – whether it is an ionic or covalent compound. Not only this, we learn about carbon dioxide when we study the carbon cycle and the photosynthesis of plants.Īdding to this, who hasn’t heard of dry ice? The solid form of CO2 that we use as a refrigerant. Other than that, we also identify its role in climate change since it is a greenhouse gas. We already know the role of CO2 in respiration. It is one of the most important compounds and we are familiar with CO2 since childhood. CO2 is the chemical formula of the molecular compound of Carbon Dioxide.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
